17 Types of Human Papillomavirus (16/18/6/11/44 Typing) Nucleic Acid Detection Kit (Fluorescence PCR)
Product name
17 Types of Human Papillomavirus (16/18/6/11/44 Typing) Nucleic Acid Detection Kit (Fluorescence PCR)
Epidemiology
Human Papillomavirus (HPV) belongs to the Papillomaviridae family of a small-molecule, non-enveloped, circular double-stranded DNA virus, with a genome length of about 8000 base pairs (bp). HPV infects humans through direct or indirect contact with contaminated items or sexual transmission. The virus is not only host-specific, but also tissue-specific, and can only infect human skin and mucosal epithelial cells, causing a variety of papillomas or warts in human skin and proliferative damage to reproductive tract epithelium.
Cervical cancer is one of the most common malignant tumors in the female reproductive tract. It has been shown that persistent HPV infection and multiple infections are one of major causes of cervical cancer. Currently there is still a lack of generally accepted effective treatments for cervical cancer caused by HPV. Therefore, early detection and prevention of cervical infection caused by HPV are the keys to prevention of cervical cancer. The establishment of a simple, specific and rapid etiological diagnosis method is of great significance for the clinical diagnosis of cervical cancer.
Technical Parameters
| Storage | ≤ -18℃ |
| Shelf-life | 12 months |
| Specimen Type | Urine sample, Female cervical swab sample, Female vaginal swab sample |
| CV | <5.0% |
| LoD | 300 Copies/mL |
| Specificity | There is no cross reaction with other reproductive tract related pathogens and human genomic DNA. |
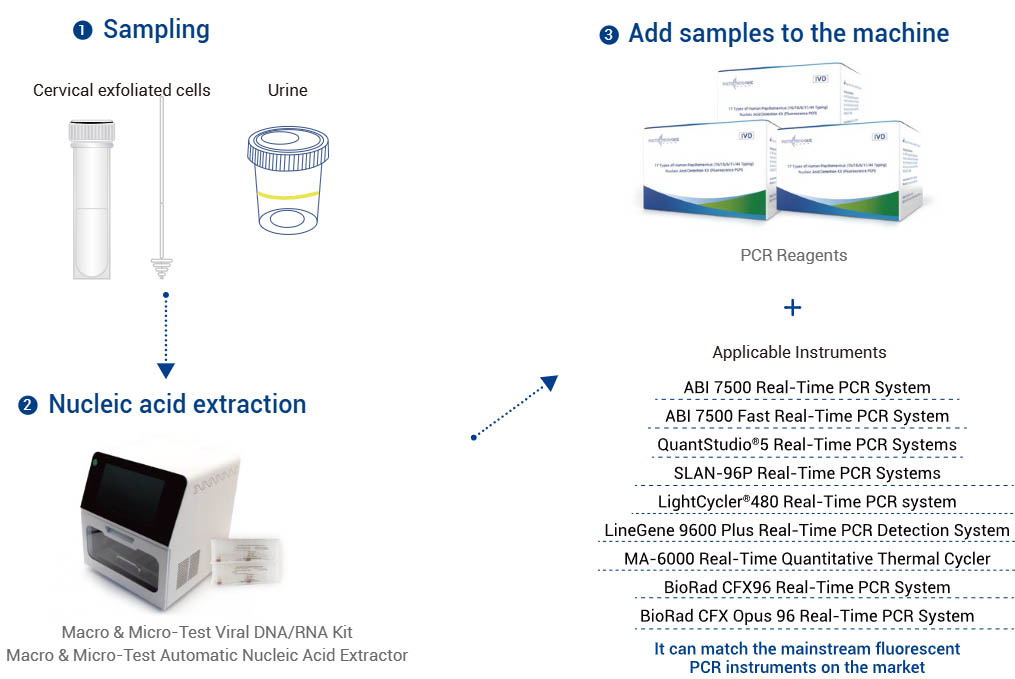
| Applicable Instruments | Applied Biosystems 7500 Real-Time PCR Systems, Applied Biosystems 7500 Fast Real-Time PCR Systems, QuantStudio®5 Real-Time PCR Systems, SLAN-96P Real-Time PCR Systems (Hongshi Medical Technology Co., Ltd.), LightCycler®480 Real-Time PCR Systems, LineGene 9600 Plus Real-Time PCR Detection Systems (FQD-96A, Hangzhou Bioer technology), MA-6000 Real-Time Quantitative Thermal Cycler (Suzhou Molarray Co., Ltd.), BioRad CFX96 Real-Time PCR System and BioRad CFX Opus 96 Real-Time PCR System. |
Work flow: See the Kit IFU for details
Solution 1 (Automated nucleic acid extraction)
Solution 2: (Macro & Micro-Test Sample Release Reagent)

Reagents need but not provided
Macro & Micro-Test Sample Release Reagent (HWTS-3005-8), Macro & Micro-Test Viral DNA/RNA Kit (HWTS-3017) (which can be used with Macro & Micro-Test Automatic Nucleic Acid Extractor (HWTS-EQ011)) by Jiangsu Macro & Micro-Test Med-Tech Co., Ltd., Macro & Micro-Test Viral DNA/RNA Column (HWTS-3020-50).
Consumables need but not provided
1.5mL of DNase/RNase free centrifuge tubes, DNase/RNase free Tips, 8-tube strips for PCR, desktop centrifuge, desktop vortex mixer.






