Since the COVID-19 pandemic, the seasonal patterns of respiratory infections have shifted. Once concentrated in the colder months, outbreaks of respiratory illness are now occurring throughout the year — more frequent, more unpredictable, and often involving co-infections with multiple pathogens.
Hospitals and clinics are reporting not only a rise in total case numbers but also more severe and complex presentations. The list of culprits is long: COVID-19, influenza A and B, RSV, adenoviruses, rhinoviruses, parainfluenza, hMPV, human bocavirus, and bacterial pathogens such as Mycoplasma pneumoniae, Chlamydophila pneumoniae, and Streptococcus pneumoniae.
Clinical Diagnosis Is More Challenging Than Ever
These pathogens frequently produce overlapping symptoms — fever, cough, sore throat, and fatigue — making them nearly indistinguishable through clinical assessment alone. In pediatric cases, RSV, hMPV, and HBoV often cause severe wheezing and bronchiolitis, while in adults, Mycoplasma pneumoniae may present with a persistent cough. COVID-19, influenza, and bacterial pneumonia can all cause high fevers and systemic symptoms, but with significant variation across age groups.
The clinical implications of misdiagnosis or delayed diagnosis are serious. Inappropriate antibiotic use, delayed antiviral treatment, ineffective isolation protocols, and misallocated resources all stem from uncertainty in etiology. And with many infections now occurring outside traditional “flu season,” relying on seasonal assumptions is no longer viable.
The Market Demands Faster, Smarter, Broader Testing
Medical laboratories, hospitals, and public health institutions are shifting their procurement priorities.
What they now need are:
-Rapid turnaround tools to support fast clinical decision-making.
-Multiplex capability to detect multiple pathogens in a single test.
-High throughput and automation to relieve pressure on staff and infrastructure.
-Stable reagents and minimal operational complexity to expand access to diagnostics in remote, emergency, or resource-limited settings.
This shift represents a growing market opportunity for distributors and diagnostic solution providers who can deliver reliable, cost-effective respiratory testing platforms.
Introducing the Eudemon™ AIO800 + 14-Pathogen Combined Detection Kit (Fluorescence PCR) (NMPA, CE, FDA, SFDA approved)
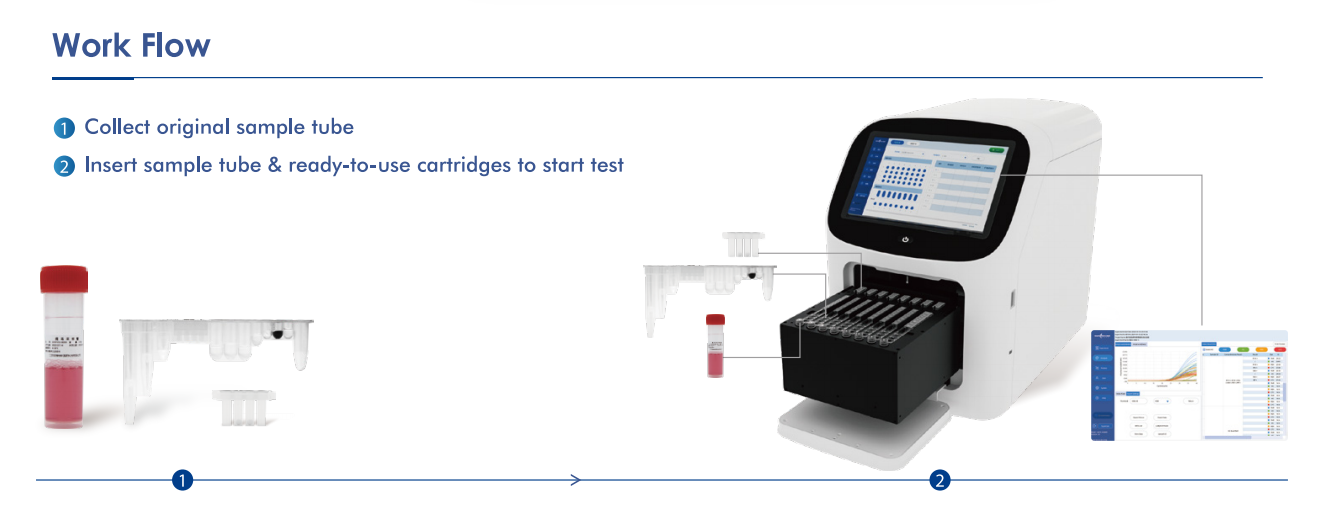
To meet this demand, the Eudemon™ AIO800 Fully Automatic Nucleic Acid Detection System, combined with a 14-pathogen respiratory panel, offers a transformative solution — delivering true “sample in, answer out” diagnostics in just 30 minutes.
This comprehensive respiratory test detects both viruses and bacteria from a single sample, enabling frontline healthcare providers to make confident, timely, and targeted treatment decisions.
Key System Features That Matter to Your Clients
Fully Automated Workflow
Less than 5 minutes hands-on time. No need for skilled molecular staff.
- Fast Results
Turnaround time of 30 minutes supports urgent clinical settings.
- 14 Pathogen Multiplex Detection
Simultaneous identification of:
Viruses:COVID-19,Influenza A & B,RSV,Adv,hMPV, Rhv,Parainfluenza types I-IV, HBoV,EV, CoV
Bacteria:MP,Cpn,SP
-Lyophilized Reagents Stable at Room Temperature (2–30°C)
Simplifies storage and transportation, eliminating cold-chain dependency.
Robust Contamination Prevention System
Includes UV sterilization, HEPA filtration, and closed-cartridge workflow, etc.
Adaptable Across Settings
Ideal for hospital labs, emergency departments, CDCs, mobile clinics, and field operations.
A Strategic Choice for Procurement and Distribution
For procurement managers, the Eudemon™ AIO800 offers not only diagnostic accuracy and efficiency but also logistical advantages that reduce operational risk and cost.
For distributors, the system’s compact design, room-temperature reagents, and minimal training requirements make it highly adaptable and scalable across a wide range of clinical environments — from tertiary hospitals to rural health centers.
In a market increasingly defined by speed, flexibility, and reliability, this solution empowers your network with a competitive, future-ready diagnostic platform.

Contact us at marketing @mmtest.com about Eudemon™ AIO800 for detailed specifications and distributor programs.
Now is the time to bring respiratory testing into a new era — with speed, clarity, and confidence.
Post time: Aug-28-2025