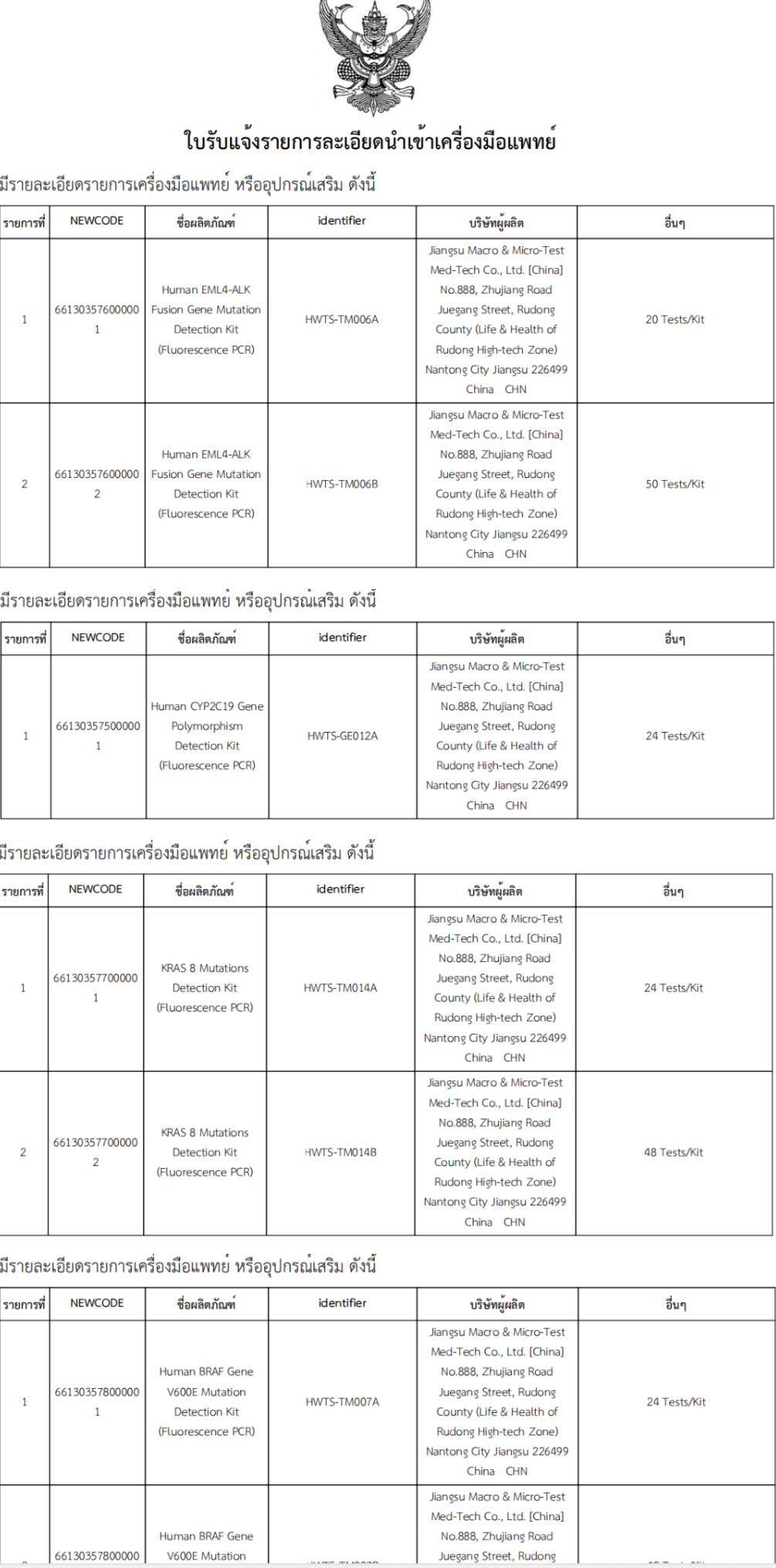
Recently, Jiangsu Macro & Micro-Test Med-Tech Co.,Ltd. "Human EML4-ALK Fusion Gene Mutation Detection Kit (Fluorescence PCR) ,Human CYP2C19 Gene Polymorphism Detection Kit(Fluorescence PCR), Human KRAS 8 Mutations Detection Kit (Fluorescence PCR) and Human BRAF Gene V600E Mutation Detection Kit (Fluorescence PCR)" were successfully approved by TFDA of Thailand!
This major breakthrough marks that the products of Macro & Micro-Test have once again won wide recognition and praise in the international market!
These kits use fluorescence PCR , which has the characteristics of high sensitivity, high specificity and simple operation, and can quickly and accurately detect the mutation of related genes, providing strong support for clinical diagnosis and treatment.
The successful approval of these products is not only an affirmation of Macro & Micro-Test technical strength and research and development ability, but also an important driving force for the company's future development!
Macro & Micro-Test has been committed to the research and development in the field of biomedicine, adhering to the concept of "people-oriented, scientific and technological innovation" and constantly introducing high-quality and high-efficiency products and services.
Thanks to TFDA of Thailand for its recognition and support of Macro & Micro-Test products, and also thanks to our customers and partners for their trust and support. We will continue to work hard, innovate and make more contributions!
Post time: Dec-01-2023
