1 Dengue Epidemic Background: An Escalating Global Public Health Challenge
Dengue is an acute mosquito-borne viral disease caused by the dengue virus (DENV), which has emerged as the most rapidly spreading arboviral disease globally, posing a substantial threat to public health security. Over the past two decades, the global incidence of dengue has increased dramatically, with the number of reported cases doubling annually since 2021 [1]. In December 2023, the World Health Organization (WHO) declared a global dengue emergency to strengthen coordinated international response efforts. Epidemiological estimates from WHO indicate that approximately 3.9 billion individuals worldwide are at risk of dengue infection, with an estimated 390 million infections occurring annually—of which 96 million present as clinically apparent cases [1,2].
2 Epidemiological Highlights
The epidemiological characteristics of dengue are shaped by the interaction of virological factors, vector ecology, host immune responses, and socio-environmental conditions. A comprehensive understanding of these characteristics is essential for the development of effective prevention and control strategies as well as accurate diagnostic approaches.
2.1 Transmission Vectors and Urban Transmission Patterns
Dengue virus is primarily transmitted by Aedes aegypti and Aedes albopictus mosquitoes. Among these vector species, Aedes aegypti is recognized as the most critical transmission vector, characterized by high “human adaptability” and widespread distribution in tropical and subtropical urban environments. Unlike other mosquito vectors of arboviral pathogens, Aedes aegypti exhibits the following key epidemiological characteristics:
-A preference for breeding in anthropogenic environments (e.g., water storage containers, discarded tires)
-A strong tropism for human blood as a nutrient source
-Daytime feeding behavior
These traits define dengue as a typical ”urban infectious disease,” with transmission efficiency significantly elevated in densely populated areas. WHO-related studies have demonstrated that in high-density urban settings, increased frequency of mosquito-human contact can substantially elevate the basic reproduction number (R₀) of DENV, thereby accelerating the spread of epidemics [2].
2.2 Global Spread Trends and Driving Factors
According to WHO reports, the global number of reported dengue cases has increased exponentially over the past two decades [1,3]. This upward trend is primarily driven by the following interconnected factors:
(1) Climate Change: Rising global temperatures not only expand the geographic range of suitable habitats for mosquito vectors but also shorten the extrinsic incubation period of DENV within the mosquito host, thereby enhancing transmission efficiency. Climate-induced variations in mosquito density have been validated by WHO as a reliable predictor of the spatiotemporal dynamics of dengue outbreaks.
(2) Urbanization: Rapid and unplanned urban expansion has created abundant breeding habitats for mosquito vectors, while increased population density has strengthened the continuity of DENV transmission chains .
(3) Global Population Movement: International travel and trade have facilitated the rapid trans-boundary transmission of DENV, promoting the transition from imported cases to sustained local transmission. WHO surveillance data show that between 2010 and 2021, the United States reported 7,528 travel-associated dengue cases, among which 3,135 required hospitalization and 19 resulted in mortality .
(4) Vector Distribution Expansion: Globally, the geographic range of Aedes aegypti and Aedes albopictus continues to expand, with Aedes mosquitoes becoming increasingly established in parts of Europe. Consequently, dengue has evolved from a traditionally regional epidemic to a global public health threat .
2.3 Multi-Serotype Co-circulation and Reinfection Mechanisms
Dengue virus comprises four antigenically distinct serotypes (DENV-1 to DENV-4). Infection with one serotype confers long-term protective immunity against that specific serotype but only temporary and partial cross-protection against the other three serotypes. The general population is universally susceptible to DENV, with only a subset of infected individuals developing clinical illness [2].
In endemic regions, multiple DENV serotypes often co-circulate simultaneously, resulting in the potential for individuals to experience multiple dengue infections throughout their lifetime. WHO epidemiological studies have identified multi-serotype co-circulation as a key driver of periodic dengue outbreaks [1].
2.4 Secondary Infection and Antibody-Dependent Enhancement
A critical and unique phenomenon in dengue epidemiology is antibody-dependent enhancement (ADE). During secondary infection with a heterologous DENV serotype, non-neutralizing antibodies produced during the primary infection facilitate viral entry into monocytes and macrophages, thereby enhancing viral replication. This mechanism is widely recognized by WHO as a major pathogenic factor in severe dengue, including dengue hemorrhagic fever and dengue shock syndrome [1].
WHO epidemiological data consistently demonstrate that individuals with secondary dengue infection have a significantly higher risk of developing severe disease compared to those with primary infection—a characteristic that is of great significance for disease surveillance and clinical management. It is important to note that while the risk of severe disease is elevated during secondary infection, infection with any DENV serotype can potentially progress to severe dengue [1].
2.5 Non-Specific Clinical Manifestations and Misdiagnosis Risk
The clinical manifestations of dengue are notably non-specific, particularly in the early stages of illness, often overlapping with those of other mosquito-borne viral infections (e.g., chikungunya and Zika viruses) as well as certain respiratory infections. WHO estimates indicate that 40-80% of DENV infections are asymptomatic [3].
Typical clinical manifestations include:
-Acute fever (persisting for 2-7 days, which may be biphasic)
-Severe headache and retro-orbital pain (pain behind the eyes)
-Muscle and joint pain (commonly referred to as “breakbone fever”)
-Macular or maculopapular rash
-Mild hemorrhagic manifestations (e.g., ecchymosis, epistaxis, gingival bleeding)
Symptomatic dengue is typically divided into three distinct phases: the febrile phase, the critical phase, and the recovery phase. Approximately less than 5% of symptomatic patients progress to severe dengue. Due to the lack of specific clinical features, diagnosis based solely on clinical symptoms is challenging, which increases the risk of misdiagnosis and underdiagnosis . The WHO explicitly emphasizes that clinical diagnosis alone is insufficient to ensure accuracy, making laboratory confirmation indispensable [1].
3 Key Points from WHO “Laboratory Testing for Dengue Virus: Interim Guidance, April 2025″
In April 2025, the World Health Organization released updated interim guidance on laboratory testing for DENV, providing authoritative technical guidance for global dengue diagnosis. This guidance synthesizes the latest evidence on dengue laboratory testing in the context of the ongoing global dengue emergency and offers practical recommendations tailored to settings with varying resource levels.

3.1 Fundamental Principles of Testing Strategy
The guidance emphasizes that dengue diagnosis must adopt a multi-marker combined testing strategy based on the stage of illness [1]. Given the absence of a universal diagnostic algorithm, testing strategies should be adapted to local epidemiological contexts, taking into account the following key factors [1]:
-Infection stage: The number of days post-symptom onset determines the most appropriate testing method
-Sample type: The suitability of whole blood, plasma, or serum for DENV detection
-Regional epidemiology: The locally circulating DENV serotypes and co-circulation of other arboviruses
-Co-infection risk: In regions with overlapping arbovirus circulation, multiplex testing should be considered to distinguish between different pathogens
3.2 Stage-Based Testing Strategy
According to the WHO guidance, dengue laboratory testing should follow clear time windows based on the stage of illness [1,2]:
(1) Acute Phase Testing (≤7 days post-onset)
-Nucleic Acid Testing (Molecular Testing): Reverse transcription-polymerase chain reaction (RT-PCR) and other molecular methods detect DENV RNA with high sensitivity.
-Antigen Testing: NS1 antigen detection, which becomes detectable within 1-3 days post-onset.
During the acute phase, viremia levels are relatively high, and nucleic acid and antigen testing achieve optimal sensitivity .
(2) Convalescent Phase Testing (≥4 days post-onset)
-Serological Testing: IgM antibodies typically become detectable around day 4 post-onset.
-In most cases, IgM antibodies persist for 14-20 days, and in some instances, persistence can extend up to 90 days.
-IgG testing has limited value for acute dengue diagnosis due to potential cross-reactive antibodies from prior flavivirus infection or vaccination.
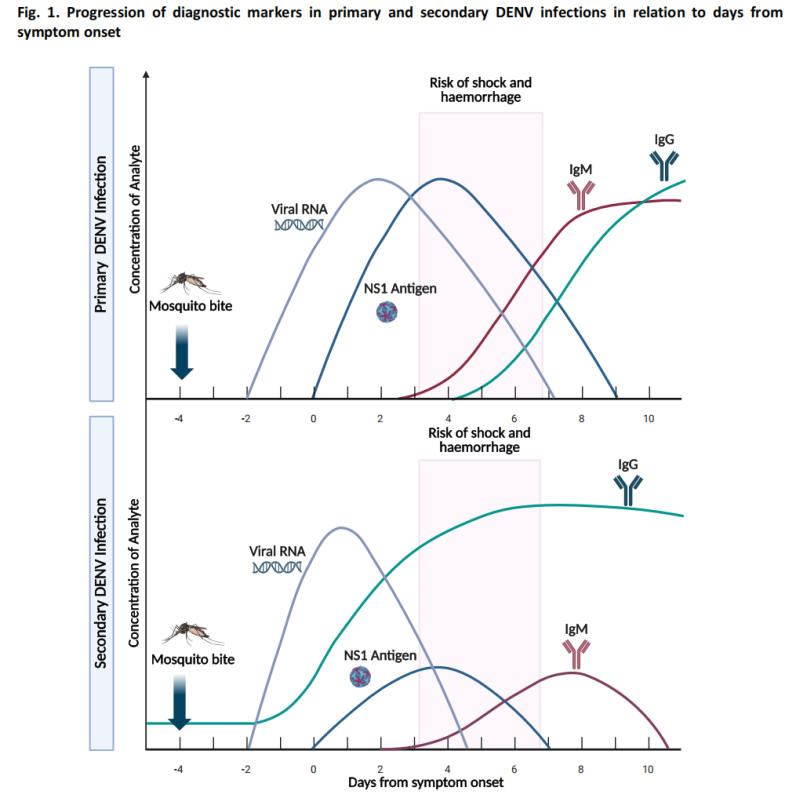
(3) Suspected Case Diagnostic Algorithm
The guidance includes a diagnostic algorithm for suspected dengue cases, recommending appropriate testing methods based on days post-symptom onset: NS1 antigen testing and nucleic acid testing are the primary approaches in the early phase, while serological testing is the primary method in the later phase.
3.3 Test Method Performance Evaluation and Selection
According tho the WHO, a systematic evaluation of the performance and applicable scenarios of various dengue testing are as following:
|
Testing Method |
Target |
Time Window |
Primary Application Scenario |
Considerations |
|
Nucleic Acid Testing |
Viral RNA | 1-7 days post-onset | Early confirmation, serotype identification | Gold standard method; requires specialized laboratory equipment and technical expertise |
|
NS1 Antigen Testing |
Non-structural protein | 1-3 days post-onset | Early rapid screening | Available in rapid diagnostic test (RDT) format, suitable for resource-limited settings |
|
IgM Antibody Testing |
Specific IgM antibodies | ≥4 days post-onset | Diagnosis of recent infection | A single serum sample only suggests a possible recent infection; seroconversion is required for confirmation |
|
IgG Antibody Testing |
Specific IgG antibodies | Convalescent/prior infection | Epidemiological investigation, assessment of immunity status | A single serum sample is not suitable for acute dengue diagnosis |
|
Combined Testing (NS1+IgM/IgG) |
Antigen + Antibodies | Full disease course | Comprehensive diagnosis of dengue infection | Currently the best-performing RDT format for dengue diagnosis |
|
NGS |
Viral RNA | 1-7 days post-onset | Viral genomic surveillance | Requires specialized sequencing equipment and bioinformatics analysis capabilities |
4 Macro & Micro-Test Dengue Detection Product Recommendations by Scenario
To support dengue prevention and control, Macro & Micro-Test provides an integrated diagnostic portfolio covering rapid screening, molecular confirmation, and genomic surveillance, meeting needs across different outbreak management stages.
4.1 Scenario 1: Rapid Screening and Targeted Surveillance
Applicable to fever clinics, primary healthcare facilities, outbreak community screening, and port/border quarantine.
-Dengue Virus NS1 Antigen Rapid Test: Detects early infection (1-3 days post-onset) with 15-minute results for rapid triage.
-Dengue Virus IgM/IgG Antibody Test: Distinguishes primary/secondary infections to evaluate severe disease risk.
-Dengue Virus NS1 Antigen + IgM/IgG Combined Rapid Test: Simultaneously detects antigen and antibodies for full-course diagnosis.
-Chikungunya Virus IgM/IgG Antibody Test: Enables differential diagnosis with dengue to identify pathogens accurately.
4.2 Scenario 2: Precision Diagnosis and Emergency Response
-Dengue Virus I/II/III/IV Nucleic Acid Detection Kit: Detects and differentiates 4 serotypes (detection limit 500 copies/mL) for outbreak tracing.
-Lyophilized Dengue Virus PCR Kit: Room-temperature transportable, suitable for resource-limited areas and sudden outbreaks.
-Dengue/Zika/Chikungunya Multiplex Real-Time PCR Kit: Simultaneously detects 3 arboviruses for efficient differential diagnosis in complex outbreaks.

All above reagents are compatible with the AIO 800 Fully Automated Sample-to-Answer System, reducing manual operation and cross-contamination, and improving efficiency and biosafety.
4.3 Scenario 3: Genomic Surveillance and Viral Lineage Analysis
Applicable to national reference laboratories, public health research institutions, aligning with WHO’s positioning of NGS.
Macro & Micro-Test’s genomic surveillance solutions support whole-genome sequencing for virus tracing, transmission chain clarification, variant monitoring, and vaccine strategy adjustment. They support manual/automated workflows, improving throughput and reproducibility, enabling laboratories to upgrade from routine testing to advanced surveillance, consistent with WHO’s emphasis on strengthening viral evolution monitoring.

4.4 Value of Integrated Solutions
Macro & Micro-Test provides complete diagnostic solutions for arbovirus detection, supporting each stage of outbreak management: rapid screening tools for frontline healthcare settings, molecular confirmation for precision diagnosis, and whole-genome analysis capabilities for epidemiological surveillance. With high-performance assays, flexible workflows, and automation-ready platforms, these solutions empower laboratories and public health systems to strengthen preparedness and response to emerging arboviral threats worldwide.
References
[1] World Health Organization. Laboratory Testing for Dengue Virus: Interim Guidance, April 2025. Geneva: World Health Organization; 2025.
[2] WHO Global Arbovirus Initiative Technical Advisory Group. Strengthening Global Preparedness and Response to Arboviral Disease Threats: A Call to Action. Lancet Infect Dis. 2026;26(1):15-17.
[3] The Lancet Microbe. Overcoming the Dilemma of Dengue Diagnostics. Lancet Microbe. 2025;6(7):101190.
Post time: Mar-20-2026
