March 24, 2026 marks the 31st World Tuberculosis Day. The World Health Organization (WHO) has announced this year’s global theme as “Yes! We can end TB!”, emphasizing that strong government leadership, sustained political commitment, and coordinated multisectoral action are essential to ending the tuberculosis (TB) epidemic.

Global Progress and Remaining Challenges
According to the Global Tuberculosis Report 2025, global TB control achieved a significant milestone in 2024, with both incidence and mortality declining for the first time since the COVID-19 pandemic.
An estimated 10.7 million people developed TB in 2024, including 54% males, 35% females, and 11% children and adolescents. Among these cases, approximately 619,000 (5.8%) were co-infected with HIV, and 390,000 (3.6%) were multidrug-resistant or rifampicin-resistant TB (MDR/RR-TB).
TB caused approximately 1.23 million deaths in 2024, remaining the leading infectious cause of death globally, surpassing COVID-19. Following three years of increases between 2021 and 2023, global TB incidence declined by nearly 2% in 2024, reflecting a gradual recovery of TB services.[1]
Geographically, 67% of cases were concentrated in eight countries: India, Indonesia, Philippines, China, Pakistan, Nigeria, Democratic Republic of the Congo, and Bangladesh.
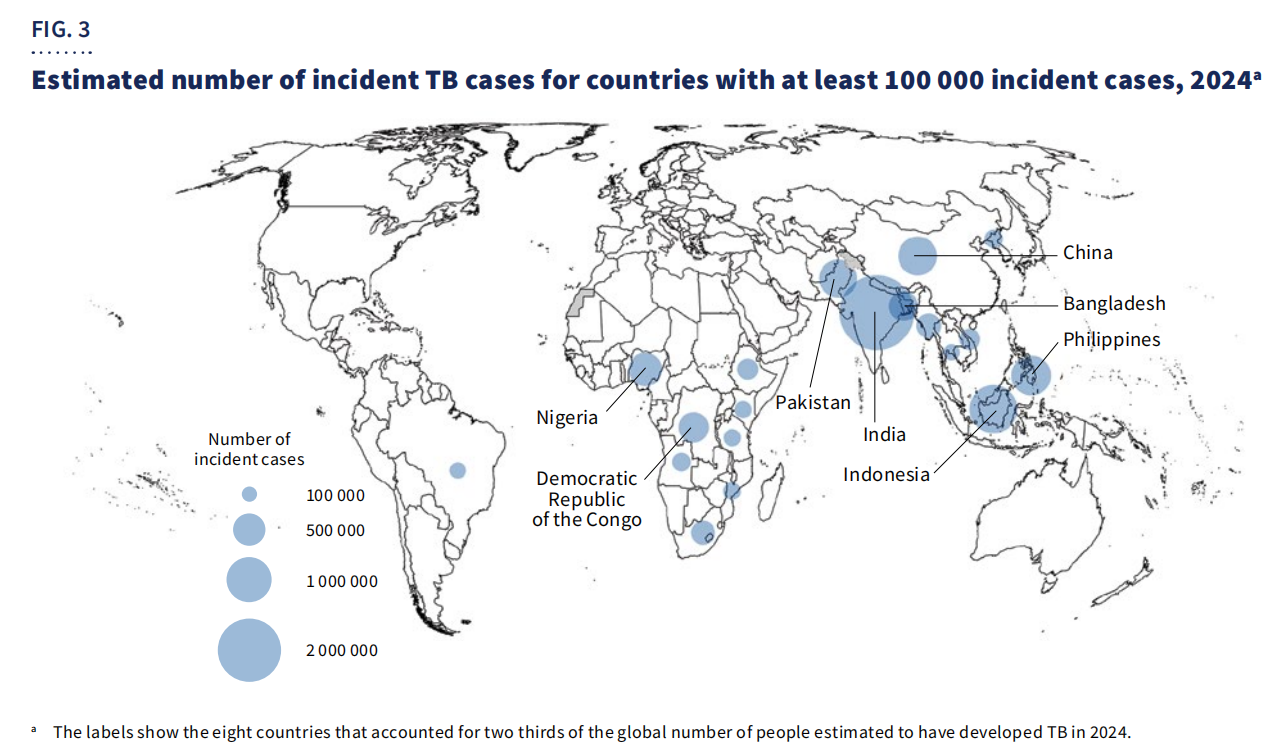
Despite progress, TB remains a leading cause of death among people living with HIV and a major contributor to mortality associated with antimicrobial resistance. Global funding continues to fall short, with only US$5.9 billion available in 2024—far below the US$22 billion annual target set for 2027.
These figures highlight the urgent need to strengthen TB control programs globally, with a focus on expanding access to diagnostics, improving treatment outcomes, and addressing the social determinants that drive TB transmission. TB continues to require robust international cooperation and political commitment to meet the goals of the End TB Strategy
Mycobacterium tuberculosis: Pathogenesis and Classification
Mycobacterium tuberculosis (MTB/M. tuberculosis) is the primary and most common pathogen responsible for tuberculosis (TB). It can enter the human body through the respiratory tract, digestive tract, or damaged skin and mucous membranes, infecting multiple organs and causing different forms of TB. Pulmonary TB, primarily transmitted through droplets, accounts for more than 80% of TB cases. Common symptoms include coughing, sputum production, and hemoptysis. After infection in the lungs, the bacteria may spread through the bloodstream to multiple systems, potentially leading to skeletal, urinary, or gastrointestinal TB.[2]
MTB is part of the genus Mycobacterium, which includes:
- Mycobacterium tuberculosis complex (MTBC): Comprising M. tuberculosis, M. bovis, M. africanum, M. canettii, and M. microti, among others. While M. tuberculosis is the primary cause of TB, M. bovis and M. africanum can also induce the disease.
- Non-tuberculous mycobacteria (NTM).
- Mycobacterium leprae, the cause of leprosy.
Laboratory Diagnostic Approaches
Accurate and timely diagnosis is essential for effective TB control. The World Health Organization emphasizes that rapid molecular diagnostic technologies have fundamentally transformed TB detection by enabling highly sensitive and specific identification of pathogens while simultaneously detecting drug resistance [1].
- Microscopy and Culture: Culture remains the gold standard for TB diagnosis, as it allows definitive identification of viable organisms and supports drug susceptibility testing and genomic analysis. However, due to the slow growth rate of Mycobacterium tuberculosis, results typically require 2–8 weeks, limiting its clinical utility in urgent decision-making.
-Immunological Testing: Immunological methods, including the tuberculin skin test (TST) and interferon-gamma release assays (IGRAs), detect host immune responses to TB infection. While useful for identifying latent infection, these assays cannot reliably distinguish between active and prior infection and therefore have limited diagnostic specificity in high-burden settings.
-Molecular Diagnostics (NAAT): DNA-based tests such as nucleic acid amplification (NAAT) are recommended for their high sensitivity and specificity.
-Targeted Next-Generation Sequencing (tNGS): Targeted sequencing technologies provide high-resolution identification of resistance-associated mutations. WHO guidelines recommend tNGS as an advanced tool for post-diagnosis drug resistance detection, supporting precision treatment strategies [3].
-Metagenomic Next-Generation Sequencing (mNGS): Metagenomic sequencing enables unbiased detection of a broad range of pathogens without prior target selection. This approach is particularly valuable in complex or unclear clinical scenarios, including mixed infections and immunocompromised patients, where conventional diagnostics may be insufficient.
WHO further highlights that microbiological confirmation is essential for initiating appropriate therapy and improving patient outcomes, reinforcing the importance of integrating advanced molecular diagnostics into TB control programs [1].
Comprehensive Molecular Diagnostic Solutions from Macro & Micro-Test
1. Multiplex PCR Detection for TB and Drug Resistance TB
|
Product Code |
Product Name |
Certification |
| HWTS-RT001 | Mycobacterium Tuberculosis DNA Detection Kit(Fluorescence PCR) |
CE |
| HWTS-RT137 | Mycobacterium Tuberculosis Isoniazid Resistance Mutation Detection Kit (Melting Curve) |
CE |
| HWTS-RT074 | Mycobacterium Tuberculosis Nucleic Acid and Rifampicin Resistance Detection Kit (Melting Curve) |
CE |
| HWTS-RT102 | Nucleic Acid Detection Kit based on Enzymatic Probe Isothermal Amplification (EPIA) for Mycobacterium tuberculosis |
CE |
| HWTS-RT144 | Freeze-dried Mycobacterium Tuberculosis Complex Nucleic Acid Detection Kit (Enzymatic Probe Isothermal Amplification) |
CE |
| HWTS-RT105 | Freeze-dried Mycobacterium Tuberculosis DNA Detection Kit (Fluorescence PCR) |
CE |
| HWTS-RT147 | Mycobacterium Tuberculosis Nucleic Acid and Rifampicin, Isoniazid Resistance Detection Kit(Melting Curve) |
CE |
When there is a strong clinical suspicion of tuberculosis (TB), the HWTS-RT147 test is recommended for the qualitative detection of MTB infection and multi-drug-resistant tuberculosis (MDR-TB). This test identifies mutations in the rpoB gene, which lead to rifampicin (RIF) resistance, and mutations in the katG and InhA genes, which are associated with isoniazid (INH) resistance. It provides an efficient, one-time test for both MTB and MDR-TB, incorporating internal quality control to minimize false-negative results, ensuring rapid and accurate results.
2. PTNseq Targeted Sequencing for Respiratory Pathogens and Resistance Profiling
|
Product Code |
Product Name |
Spec. |
| HWKF-TS0001 | PTNseq Bloodstream Infection Pathogen Gene Enrichment Kit | 24 tests/kit |
| HWKF-TS0002 | PTNseq Central Nervous System Infection Pathogenic Microorganism Gene Enrichment Kit | 24 tests/kit |
| HWKF-TS0003 | PTNseq Respiratory Infection Pathogen Gene Enrichment Kit | 24 tests/kit |
| HWKF-AT0003 | PTNseq Respiratory Infection Pathogenic Microorganisms Automated Enrichment Library Construction Kit (ONT) | 24 tests/kit |
| HWKF-TS0004 | PTNseq Broad-Spectrum Infectious Pathogens Gene Enrichment Kit | 24 tests/kit |
| HWKF-TS0005 | PTNseq Ultra-Broad-Spectrum Infectious Pathogenic Microorganism Gene Enrichment Kit | 24 tests/kit |
| HWKF-TS0151 | Mycobacterium Typing and Drug Resistance Gene Enrichment Kit (Multiple Amplification Method) | 24 tests/kit |
In cases of mixed respiratory infections (including upper and lower respiratory tract infections, tuberculosis, and chronic respiratory diseases), or when drug resistance gene analysis is required (e.g., suspected drug-resistant tuberculosis), the PTNseq series of targeted high-throughput gene detection can be applied. Based on advanced targeted sequencing technology, PTNseq employs ultra-multiplex PCR to enrich specific target sequences, combined with high-throughput sequencing and third-generation nanopore technology for comprehensive pathogen identification and drug resistance profiling.
The system uses patented, high-specificity primers for ultra-multiplex amplification of target genes. Supported by a proprietary database and intelligent bioinformatics algorithms, it provides accurate pathogen identification along with drug resistance and virulence gene analysis. Targeted enrichment reduces interference from host DNA, improving sensitivity in samples with high human background and enabling effective detection of difficult targets such as Mycobacterium tuberculosis, fungi, intracellular bacteria, RNA viruses, and resistance or virulence genes.
PTNseq achieves a limit of detection as low as 100 copies/mL and covers 175 common respiratory pathogens, including 76 bacteria, 73 viruses, 19 fungi, 7 mycoplasmas, as well as Chlamydia, Rickettsia, and 54 drug resistance genes. The panel includes the Mycobacterium tuberculosis complex and major non-tuberculous mycobacteria.
The PTNseq series combines high sensitivity with cost-effectiveness, improving pathogen detection rates and supporting individualized antimicrobial therapy while helping mitigate antimicrobial resistance. Integrated with the fully automated gene sequencing library preparation system (AIOS), it provides a streamlined, in-hospital solution with a turnaround time as fast as 6.5 hours from sample to result.

3. Metagenomic Sequencing for Broad-Spectrum Pathogen Detection
|
Product Code |
Product Name |
Spec. |
|
HWKF-MN0011 |
Metagenomic Pathogen Detection Kit(DNA-Illumina) |
24 tests/kit |
|
HWKF-MN0018 |
Metagenomic Pathogen Detection Kit(DNA-MGI) |
24 tests/kit |
|
HWKF-MN0021 |
Metagenomic Pathogen Detection Kit(DNA-ONT) |
24 tests/kit |
|
HWKF-MN0012 |
Metagenomic Pathogen Detection Kit(RNA-Illumina) |
24 tests/kit |
|
HWKF-MN0019 |
Metagenomic Pathogen Detection Kit(RNA-MGI) |
24 tests/kit |
|
HWKF-MN0022 |
Metagenomic Pathogen Detection Kit(RNA-ONT) |
24 tests/kit |
|
HWKF-MN0013 |
Metagenomic Pathogen Detection Kit(DNA+RNA-Illumina) |
24 tests/kit |
|
HWKF-AYM0013 |
Metagenomic Pathogen Detection Automated Library Construction KiT (DNA+RNA-Illumina) |
24 tests/kit |
|
HWKF-MN0020 |
Metagenomic Pathogen Detection Kit(DNA+RNA-MGI) |
24 tests/kit |
|
HWKF-MN0023 |
Metagenomic Pathogen Detection Kitt(DNA+RNA-ONT) |
24 tests/kit |
When clinical diagnosis is unclear, mNGS pathogen high-throughput gene detection can be performed on various samples from the patient, including bronchoalveolar lavage fluid, sputum, throat swabs, blood, pleural effusion, pus, and tissue samples. This approach uses metagenomic sequencing technology, where different samples undergo targeted pre-treatment followed by nucleic acid extraction using glass beads and wall-digesting enzymes, enhancing extraction efficiency. Sequencing is adapted to multiple platforms, ensuring high data volume for improved mNGS sensitivity and assembly integrity. The data is analyzed using a self-built database and intelligent algorithms to detect over 20,000 pathogens, including bacteria, fungi, viruses, and parasites, providing information on suspected pathogenic microorganisms. This method is suitable for difficult-to-diagnose, critically ill, or immunocompromised patients, including the identification of MTB complex and NTM, as well as mixed infections. It significantly improves pathogen detection rates and helps guide clinical targeted antibiotic use, enabling precise infection diagnosis.
Conclusion
Although substantial progress has been achieved, tuberculosis remains a major global health challenge, particularly in the context of drug resistance, funding gaps, and unequal access to diagnostics.
WHO emphasizes that scaling up access to rapid molecular diagnostics and advanced sequencing technologies is essential to achieving the goals of the End TB Strategy. With continued innovation, investment, and global collaboration, ending TB is no longer an aspiration, but an attainable objective.
References:
- World Health Organization. Global Tuberculosis Report 2024/2025: Diagnostic Testing and Treatment.
- World Health Organization. WHO Manual for Selection of Molecular WHO-Recommended Rapid Diagnostic Tests for Detection of Tuberculosis and Drug-Resistant Tuberculosis.
- World Health Organization. WHO Consolidated Guidelines on Tuberculosis: Module 3 – Diagnosis (2024 Update).
Post time: Mar-24-2026
