1. Global Epidemiological Update of Mpox (as of March 2026)
Mpox, caused by the Monkeypox virus (MPXV), remains a persistent global public health concern despite the end of its designation as a Public Health Emergency of International Concern (PHEIC) by the World Health Organization. Continued transmission across multiple regions highlights the need for sustained surveillance and advanced diagnostic capacity.
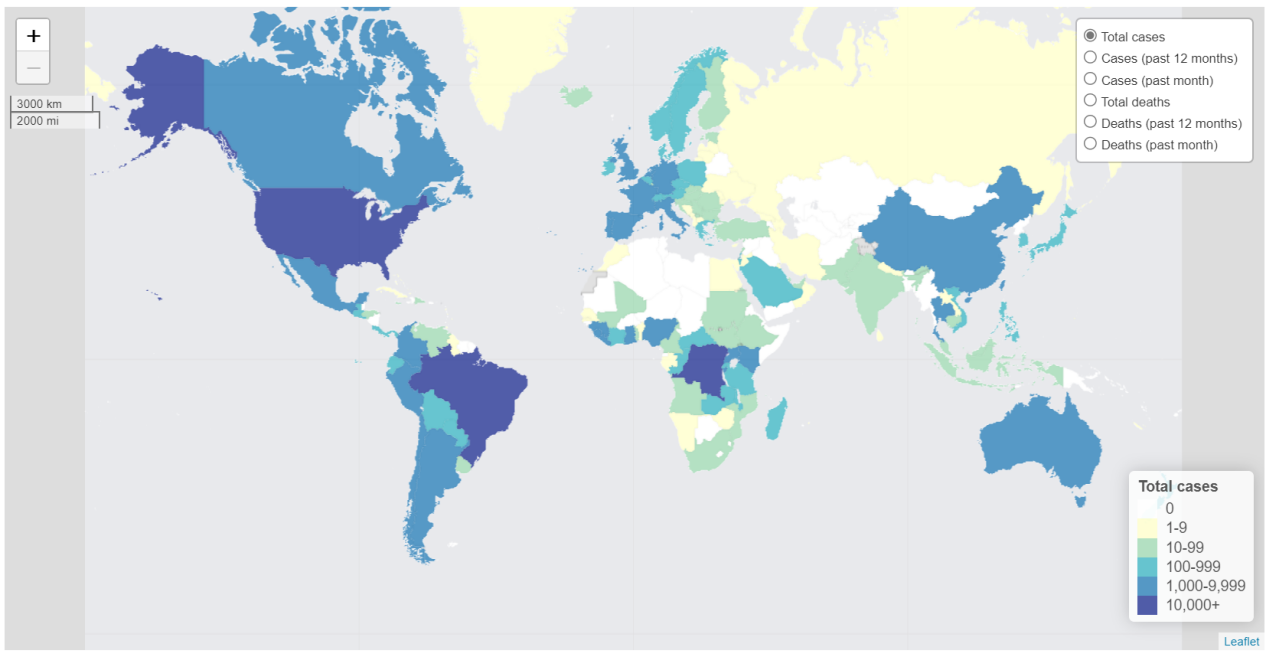
Since the multinational outbreak in 2022, nearly 128,000 laboratory-confirmed cases and 281 deaths have been reported across more than 130 countries [1]. In 2025 alone, 50,751 confirmed cases and 206 deaths were documented by 96 countries, with over 90% of cases occurring in Africa. Notably, the Democratic Republic of the Congo, Sierra Leone, and Uganda accounted for approximately 32,000 cases, underscoring the disproportionate burden in endemic regions [1].
All major MPXV clades (Ia, Ib, and II) continue to circulate. The emergence and international spread of Clade Ib—including first detections in the Czech Republic, Israel, Madagascar, and Nepal—as well as documented community transmission in France, Italy, and Spain, indicates evolving transmission dynamics [1].
2. Clinical Manifestations and Diagnostic Challenges
Mpox presents with a spectrum of clinical features, ranging from asymptomatic infection to severe systemic disease. Common symptoms include fever, headache, lymphadenopathy, myalgia, and fatigue etc. The characteristic rash evolves through macular, papular, vesicular, and pustular stages before crusting, and may involve the face, extremities, oral mucosa, and anogenital regions.
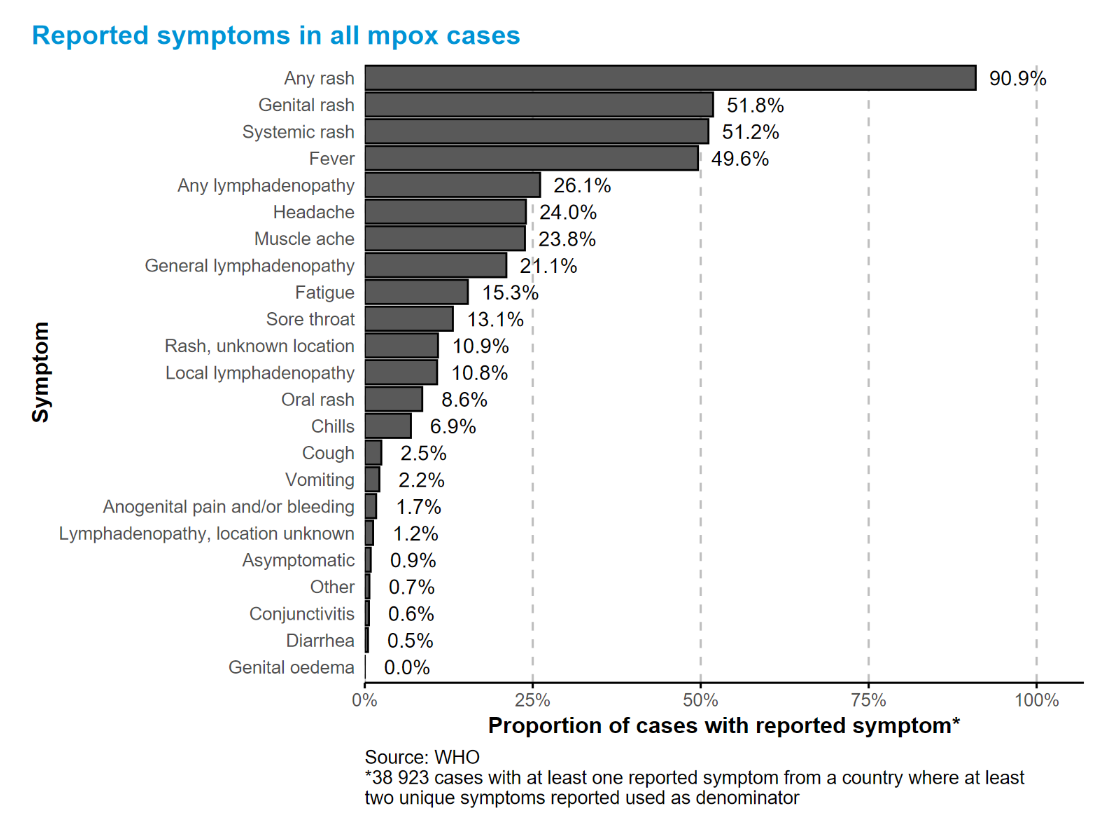
While most cases are self-limiting within 2–4 weeks, severe outcomes are more likely in vulnerable populations, including infants, pregnant women, and immunocompromised individuals (e.g., advanced HIV infection) [2].
The increasing occurrence of atypical and mild presentations, including asymptomatic cases, poses significant diagnostic challenges. This underscores the critical importance of highly sensitive, specific, and rapid diagnostics to support early detection and interrupt transmission chains.
3. Prevention, Control, and the Role of Diagnostics
Transmission of MPXV occurs through multiple routes, including direct contact with lesions or bodily fluids, respiratory droplets during prolonged close contact, sexual transmission, contaminated fomites, and vertical transmission.
Key control measures include:
- -Rapid identification and isolation of cases
- -Contact tracing and community-based surveillance
- -Risk communication and behavioral interventions
- -Vaccination for pre- and post-exposure prophylaxis
The WHO emphasizes the importance of strengthening laboratory capacity and deploying appropriate diagnostic tools tailored to different healthcare settings—from decentralized rapid testing to centralized genomic surveillance platforms [1].
4. Macro & Micro-Test: Integrated Diagnostic Solutions Driven by Technological Innovation
Against the backdrop of evolving mpox epidemiology, Macro & Micro-Test has developed a comprehensive, multi-platform diagnostic portfolio, demonstrating strong capabilities in assay design, molecular engineering, and translational application.
4.1 Rapid Immunochromatographic Assays: Accessible Point-of-Care Testing
The company provides rapid, instrument-free detection within 10–20 minutes, supporting decentralized testing and large-scale screening:
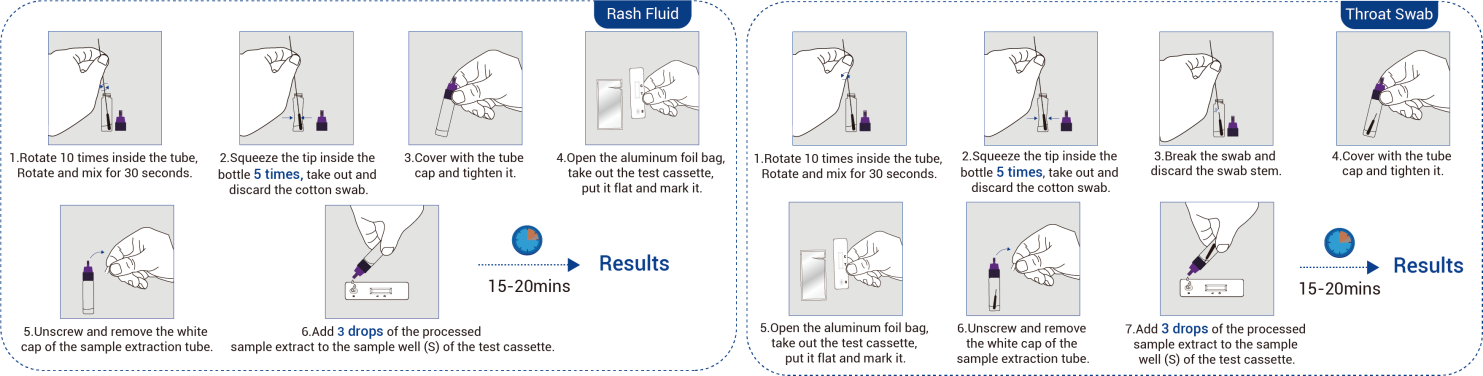
- Antigen Detection Kit (HWTS-OT079):
- Easy sampling (rash fluid/oral swab, serum)
- Limit of detection (LoD): 20 pg/mL
- Broad clade coverage (Clade I & II)
- No cross-reactivity with similar viruses
- Overall percent agreement (OPA): 96.4% vs nucleic acid amplification tests (NAATs)
- IgM/IgG Antibody Detection Kit (HWTS-OT145):
- Easy sampling (whole blood, serum, plasma)
- Identifies IgM and IgG to decide mpox infection stages
- Suitable for seroepidemiological investigations
These assays are optimized for stability (4–30°C, 24 months), addressing logistical constraints in resource-limited settings.
4.2 Isothermal Amplification (EPIA): Ultra-Rapid Molecular Detection
The EPIA-based nucleic acid detection kit (HWTS-OT200) represents a significant advancement in rapid molecular diagnostics:
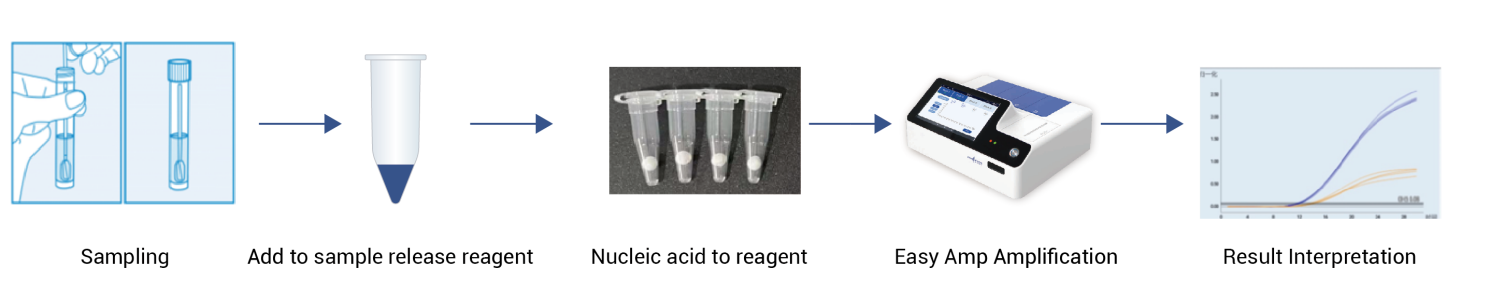
- LoD: 200 copies/mL (comparable to qPCR)
- Time-to-positive: as fast as 5 minutes
- On-demand testing enabled by independent modules of Easy Amp System
- Lyophilized formulation enabling room-temperature storage
- Clinical performance: (PPA: 100% , NPA: 100% , OPA: 100%) (Kappa = 1.000)
Importantly, the assay demonstrates no cross-reactivity with orthopoxviruses (smallpox, vaccinia, cowpox) or common herpesviruses (HSV, VZV), reflecting high analytical specificity.
4.3 Fluorescence qPCR: High-Sensitivity, WHO-Recognized Diagnostics
Macro & Micro-Test’s fluorescence PCR portfolio highlights its compliance with international standards and regulatory recognition:
- HWTS-OT071 (WHO EUL-listed):
- Dual-target design ensuring high robustness
- LoD: 200 copies/mL
- Flexible sampling of rash fluid, throat swab and serum
- High specificity without cross reactivity with smallpox virus, vaccinia virus, cowpox virus, mousepox virus, herpes simplex virus, varicella-zoster virus, and human genome, etc.
- Clinical performance: PPA 100%, NPA 99.40%, OPA 99.64% vs sequencing
- Freeze-dried qPCR Kit (HWTS-OT078):
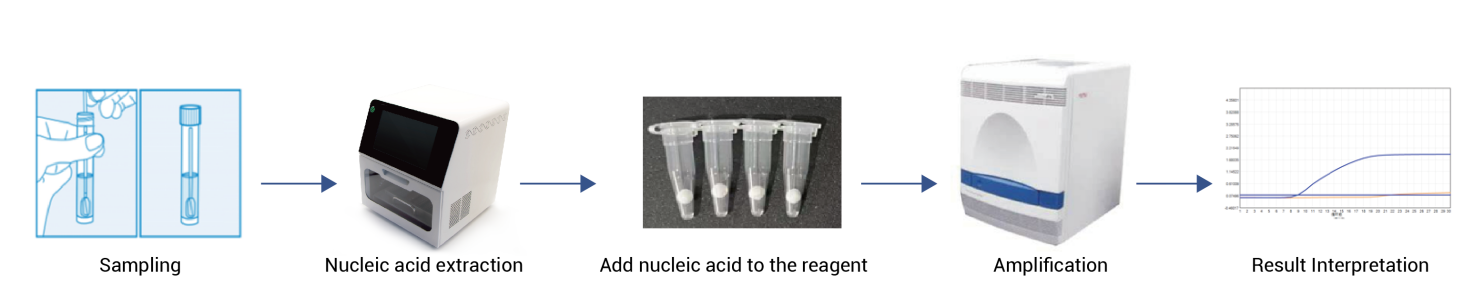
- Eliminates cold-chain dependency while maintaining performance
- Orthopox Universal Detection Kit (HWTS-OT072):
- Simultaneous detection of four human orthopox viruses (Mpox included)
- LoD: 200 copies/mL
- High specificity without cross reactivity with similar pathogens
- Reduces risk of misdiagnosis and enhances differential detection
- Monkeypox Virus Typing Kit (HWTS-OT201):
- Enables precise differentiation between Clade I and Clade II
- LoD: 200 copies/mL
- Critical for epidemiological tracking and outbreak investigation
4.4 Whole-Genome Sequencing: Enabling Genomic Surveillance and Variant Tracking
Macro & Micro-Test further demonstrates advanced capabilities in pathogen genomics through its whole-genome sequencing solutions:
- Monkeypox Virus Universal Whole Genome Kit (Multi-PCR NGS) (HWTS-TW0042):
- Turnaround time: 6–8 hours
- ≥98% genome coverage (Ct ≤32)
- Flexible compatibility with multiple 2nd and 3rd generation sequencing platforms (ONT, Qi Carbon, Salus, Illumina, MGI…)
- Ultra-sensitive Monkeypox Virus Whole Genome Detection Kits (Illumina/MGI) (HWTS-TW0041/0043):
- Detection threshold: Ct ≤35
- High-density primer design (1,448 pairs of 200bpamplicons)
- Validated with over 400 clinical samples
- Flexible compatibility with multiple 2nd generation sequencing platforms (Qi Carbon, Salus, Illumina, MGI…)
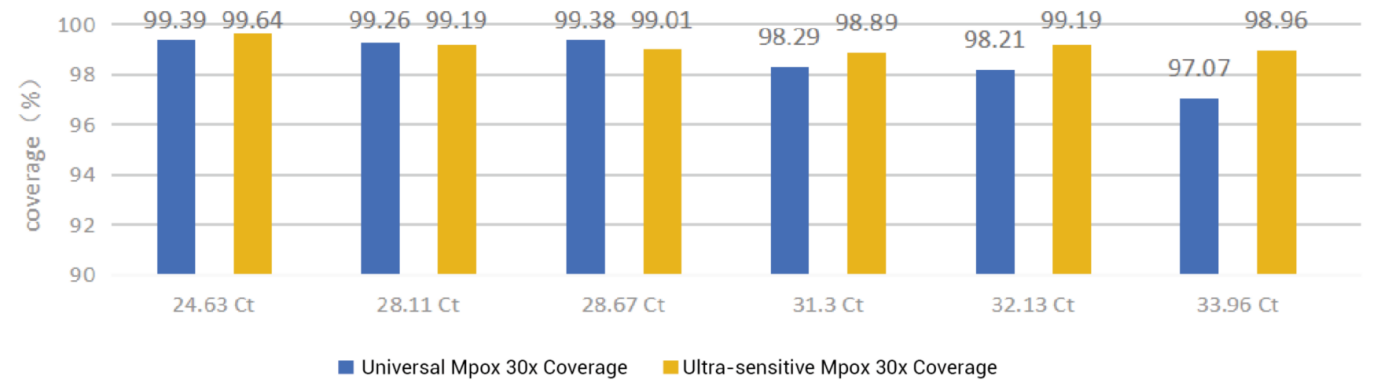
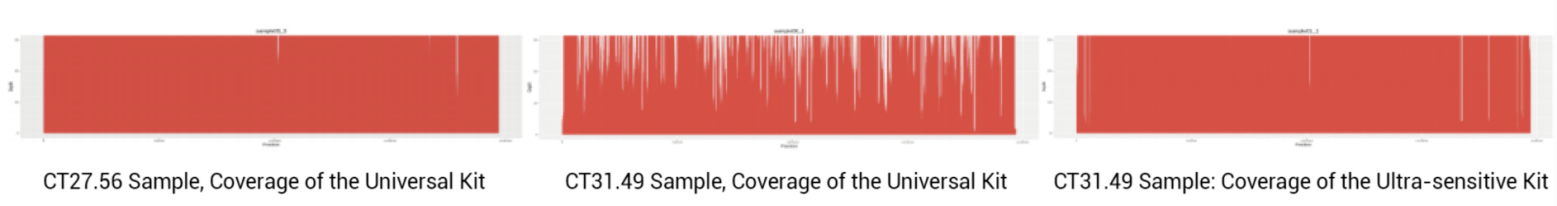
Clinical Statistics of Mpox Virus Whole Genome Sequencing
Coverage of Mpox Virus Whole Genome Sequencing
These Mpox WGS kits support high-resolution genomic epidemiology, including mutation tracking, transmission mapping, and variant evolution analysis.
5. Conclusion: Advancing Global Mpox Control Through Scientific Excellence
The continued global circulation and genetic diversification of MPXV necessitate a multi-tiered diagnostic strategy integrating rapid screening, confirmatory molecular testing, and genomic surveillance.
Macro & Micro-Test’s comprehensive diagnostic ecosystem reflects strong scientific innovation across multiple technological domains, including immunoassays, isothermal amplification, real-time PCR, and next-generation sequencing. These solutions not only address current clinical and public health needs but also align with international standards and emerging diagnostic paradigms.
By enabling rapid detection, precise clade differentiation, and real-time genomic surveillance, the company plays a critical role in strengthening global preparedness and response to mpox and other emerging infectious diseases.
References:
[1] WHO. Mpox Global Situation Reports.March, 2026, Geneva: World Health Organization; 2026
[2] WHO. Clinical Management and Infection Prevention for Mpox. May 2025, Geneva: World Health Organization; 2025
Post time: Mar-31-2026