March 4th is International HPV Awareness Day—a vital opportunity to spotlight one of the most preventable cancers affecting women worldwide: cervical cancer. Despite its global burden, cervical cancer is largely preventable through vaccination and, critically, early and accurate detection.
What Is HPV?
Human Papillomavirus (HPV) is one of the most common viral infections worldwide. Most sexually active individuals will encounter HPV at some point in their lives.
While the majority of infections clear naturally without symptoms, certain high-risk HPV types can persist in the body. Over time, persistent infection may cause abnormal cellular changes that can develop into cervical cancer if not detected and managed early.
At Macro & Micro-Test, we are committed to empowering women and healthcare providers with the tools needed to intervene early. To understand why screening is so essential, we must first address a fundamental question: How long does it take for an HPV infection to develop into cervical cancer?
The Timeline: A 10–20 Year Window of Opportunity
The progression from an initial Human Papillomavirus (HPV) infection to cervical cancer does not happen overnight. It is typically a gradual process spanning 10 to 20 years, offering a substantial window for timely medical intervention—provided effective screening is in place.
Here is a closer look at this progression:
Initial Infection (0–6 months):
HPV enters the cervical epithelial cells. In most cases, the immune system clears the virus within 6–24 months without causing lasting harm.
Persistent Infection (2–5 years):
If the immune system fails to eliminate the virus, the infection becomes persistent. Viral oncogenes (E6/E7) begin interfering with key tumor suppressor proteins, disrupting normal cellular regulation.
Cervical Intraepithelial Neoplasia (CIN) (3–10 years):
Persistent infection may lead to precancerous cellular changes graded from CIN1 to CIN3. This stage represents the critical intervention point—detecting and treating CIN can prevent progression to cancer.
Malignant Transformation (5–20 years):
If high-grade lesions (CIN3) remain untreated, they may eventually develop into invasive cervical cancer.
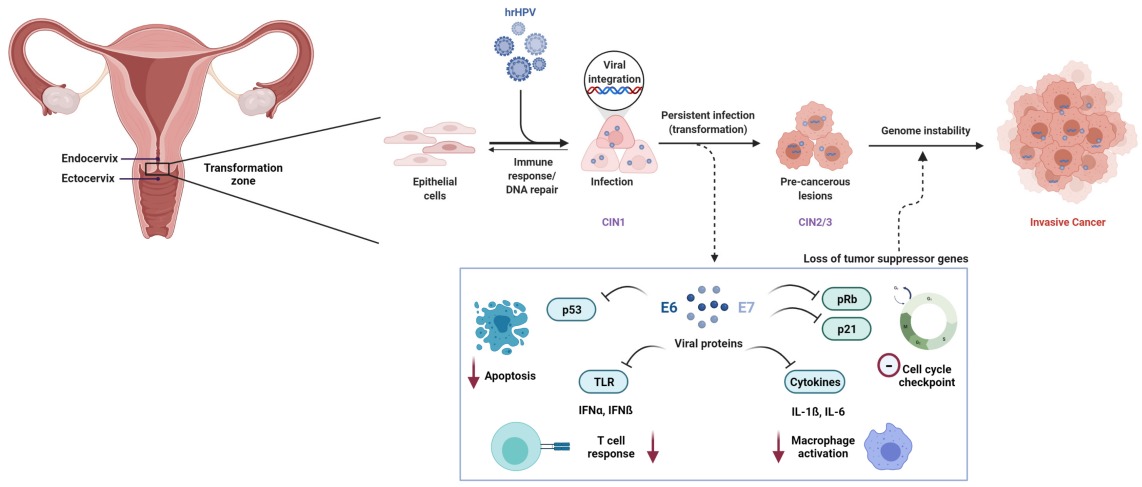
The key message: there is often a decade—or more—to detect and stop disease progression. Regular, reliable screening makes this possible.
The Importance of Early Detection: Why Screening Matters
Early-stage HPV infections and precancerous changes typically produce no symptoms. Screening is therefore the most effective defense.
While cytology (Pap smear) has played a vital role in cervical cancer prevention, modern molecular diagnostics provide earlier and more precise detection.
HPV Screening: Methods, Limitations, and Recommended Intervals
Cytology (Pap Smear): Examines cervical cells for abnormalities with moderate sensitivity, often missing early infections, and is recommended every 3 years for women aged 21–29 or every 3–5 years with HPV co-testing for women aged 30–65.
HPV DNA Testing: Highly sensitive for detecting HR-HPV DNA, ideal for broad primary screening and early HPV infections, with a recommended interval of every 5 years for women aged 25–65.
HPV mRNA Testing: Targets E6/E7 mRNA to identify infections more likely to progress, offering better risk stratification.
Macro & Micro-Test: Comprehensive Solutions for Every Screening Need
Recognizing that different clinical scenarios require different approaches, Macro & Micro-Test offers a comprehensive portfolio of HPV detection kits. From broad DNA screening and genotyping to targeted risk assessment through mRNA analysis, our solutions deliver accuracy, flexibility, and dependable performance.

Key Product Features
High Sensitivity: Detection limits as low as 300 copies/mL for DNA kits and 500 copies/mL for mRNA kits, supporting early and reliable identification of HPV infections.
Broad Platform Compatibility: Designed for seamless integration with mainstream PCR systems, enabling smooth incorporation into existing laboratory workflows.
Flexible Sample Types: Validated for cervical/vaginal swabs and urine samples, accommodating diverse clinical practices.
Comprehensive Coverage: Options ranging from 14-type high-risk screening to 28-type low & high-risk HPV DNA genotyping, as well as 15-type high-risk E6/E7 mRNA detection for risk stratification—allowing clinicians to select the most appropriate solution for screening, triage, and patient management.
Spotlight on Our Flagship HPV Solutions
For effective and more accessible primary screening, Macro & Micro-Test’s HPV high-risk 14 solutions are designed to detect the most clinically significant high-risk HPV types responsible for the cervical cancers.
As our flagship HPV products, the following two essential kits are fully compatible with the AIO 800 Fully Automated Molecular System, enabling a seamless Sample-to-Answer workflow. Integration with AIO 800 streamlines laboratory operations, reduces hands-on time, and ensures reliable results with minimal manual intervention—from sample loading to final analysis.

-Kit 1: 14 Types of HPV Nucleic Acid Typing Detection Kit (Fluorescence PCR)
Cat. No. HWTS-CC012A
The Ideal Choice for Comprehensive Genotyping
This kit detects and differentiates 14 high-risk HPV types, providing detailed genotyping information to support precise risk stratification and individualized patient management.
-Kit 2: 14 Types of High-risk Human Papillomavirus (16/18 Typing) Nucleic Acid Detection Kit (Fluorescence PCR)
Cat. No. HWTS-CC007A
The Ideal Choice for Primary Triage and Risk Assessment
HPV types 16 and 18 account for approximately 70% of cervical cancer cases worldwide. This kit detects the full panel of 14 high-risk HPV types while specifically identifying and differentiating HPV 16 and HPV 18—supporting immediate triage decisions and targeted clinical follow-up.
Both kits are available in liquid and lyophilized formats, validated for cervical/vaginal swab and urine samples, and offer a sensitivity of 300 copies/mL. When paired with the AIO 800 Fully Automated Molecular System, they provide a truly automated, efficient, and reliable HPV screening solution.
Closing the Gap Through Early Detection
The fight against cervical cancer is a race against time—but it is a race we can win. By understanding the slow progression of HPV-related disease and leveraging advanced molecular screening technologies, abnormalities can be detected and treated long before they become life-threatening.
This International HPV Awareness Day, let us move beyond awareness and take meaningful action. With Macro & Micro-Test’s comprehensive, user-friendly, and accurate HPV detection solutions, we can expand access to screening worldwide and move closer to the goal of eliminating cervical cancer.
For more information about our HPV full molecular diagnostics portfolio, please contact your local Macro & Micro-Test representative.
Post time: Mar-04-2026
