1.Global Burden of Cervical Cancer and Screening Challenges
Cervical cancer remains a major global public health challenge, despite being largely preventable through effective screening and early intervention. According to the World Health Organization (WHO), an estimated 662,000 new cases and 349,000 deaths occurred worldwide in 2022, ranking it as the fourth most common cancer and the fourth leading cause of cancer-related mortality among women. The burden of disease is disproportionately concentrated in low- and middle-income countries (LMICs), where incidence and mortality rates are significantly higher than in high-income settings. They are primarily due to a lack of high-quality screening programs and efficient detection and treatment of precancerous lesions, compounded by:
-Absent or weak screening infrastructure: Lack of accessible, quality-assured screening services.
-Resource constraints: Limited laboratory infrastructure, cold chain logistics, and reliable electricity.
-Workforce shortages: A lack of trained laboratory and clinical staff.
-Delays and loss to follow-up: Lag time between sample collection and the availability of results, leading to delays in clinical management or patients being lost to follow-up[1].
2. Etiology and Molecular Basis of Cervical Carcinogenesis
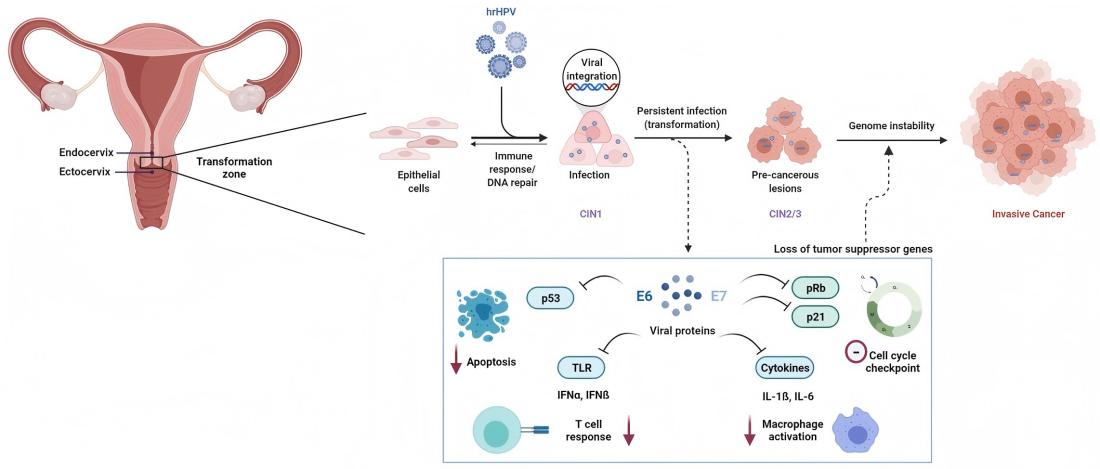
Persistent infection with high-risk human papillomavirus (HR-HPV) is a necessary cause of cervical cancer. Among more than 200 identified HPV genotypes, at least 12 types have been classified as carcinogenic (Group 1) by the International Agency for Research on Cancer (IARC).
At the molecular level, HPV-mediated carcinogenesis is primarily driven by the viral oncoproteins E6 and E7. E6 promotes the degradation of the tumor suppressor protein p53, while E7 functionally inactivates the retinoblastoma protein (Rb), resulting in dysregulation of the cell cycle and malignant transformation.
3.WHO-Recommended Screening Strategies
The World Health Organization recommends HPV nucleic acid testing (NAT) as the preferred primary screening method for cervical cancer prevention.
General population: HPV DNA or mRNA-based NATs
Women living with HIV: HPV DNA-based NATs
Screening intervals:
Women aged 30–65 years: Every 5–10 years
Women living with HIV: Every 3–5 years
Compared with cytology-based methods, HPV testing demonstrates higher sensitivity and provides a superior negative predictive value, enabling longer and more cost-effective screening intervals.
4.WHO Target Product Profile for HPV Screening Tests
The WHO has developed a Target Product Profile (TPP) for HPV screening assays intended for use in decentralized and resource-limited settings.[1]
Key characteristics include:
-Compatibility with self-collected specimens
-Detection of multiple high-risk HPV genotypes (≥12 types)
-Operation by non-laboratory-trained personnel
-Results available within a single clinical encounter
These criteria support point-of-care testing and “screen-and-treat” strategies.
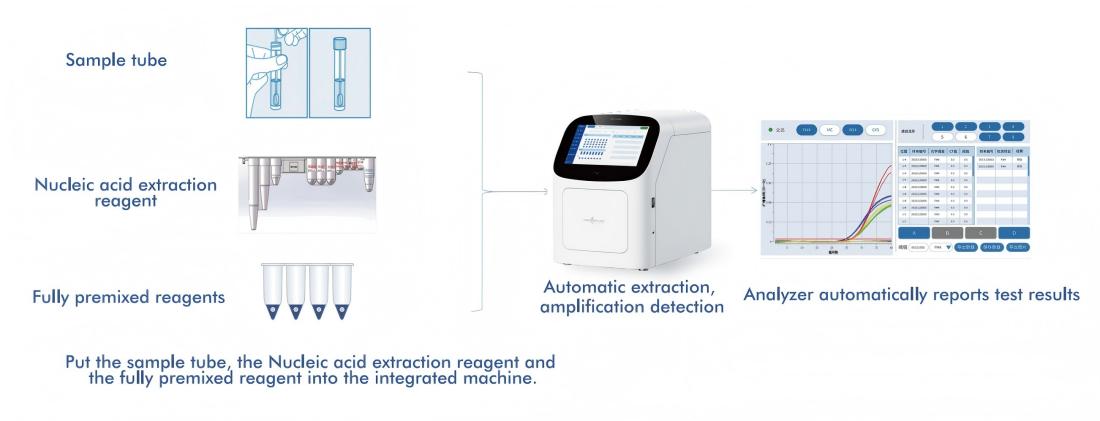
5.A Fully Automated High-Risk HPV Detection Platform
Macro & Micro-Test’s AIO800 system provides a fully automated Sample-to-answer workflow integrating nucleic acid extraction, amplification, and detection in line with WHO-recommended screening strategies.
5.1 Automated Sample-to-Answer Workflow
The platform integrates nucleic acid extraction, amplification, and detection into a single, closed system, requiring minimal operator intervention. This design:
-Reduces dependence on specialized laboratory personnel
-Minimizes process variability and contamination risk
-Enables deployment in decentralized healthcare settings
At the same time, its throughput capacity supports application in centralized laboratories, facilitating large-scale screening programs.
5.2 Broad Genotyping Coverage
The system detects 14 high-risk HPV types, including all 12 carcinogenic types classified by IARC (HPV16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, and 59), as well as HPV66 and HPV68.
Importantly, it provides type-specific genotyping results, enabling risk stratification and more precise clinical management.
5.3 Analytical Sensitivity and Clinical Implications
With the limit of detection of 300 copies/mL, the system is capable of identifying low-level HPV infection contributing to:
-Earlier detection of clinically relevant infections
-Improved negative predictive value
-Support for extended screening intervals
5.4 Support for Self-Sampling
The platform is compatible with both cervical swab samples and self-collected urine specimens aligning with WHO recommendations to address key barriers to screening uptake, including:
-Limited access to healthcare facilities
-Sociocultural constraints
-Patient discomfort with clinician-based sampling
Self-sampling has been shown to increase participation in screening programs, particularly among underserved populations.
5.5 Lyophilized Reagent Technology for Ambient Storage
To further enhance applicability in resource-limited settings, the system incorporates lyophilized (freeze-dried) reagents that are stable at ambient temperature, offering several operational advantages:
-Cold-chain independence: Eliminates the need for refrigerated transport and storage
-Improved stability: Maintains reagent integrity under variable environmental conditions
-Simplified logistics: Reduces infrastructure requirements and operational costs
-Enhanced accessibility: Facilitates deployment in remote or underserved regions
6.Conclusion
The transition from cytology-based screening to HPV-based molecular testing represents a paradigm shift in cervical cancer prevention. By targeting the underlying cause of disease rather than morphological changes, HPV testing enables earlier and more sensitive detection of at-risk individuals.
Moreover, the fully automated molecular diagnostic platforms offer a practical approach to overcoming existing barriers in screening delivery. By combining high analytical performance with operational simplicity and support for self-sampling, AIO800 has the potential to expand screening coverage, improve early detection, and contribute to reducing the global burden of cervical cancer.
References:
1. World Health Organization. Target product profiles for human papillomavirus screening tests to detect cervical precancer and cancer. Geneva: WHO; 2024.
Post time: Mar-28-2026