Once confined to the tropics, mosquito-borne viral diseases are rapidly expanding their reach. Dengue and chikungunya, sharing the same vectors, overlapping symptoms, and growing geographic footprints, are evolving from episodic outbreaks into a persistent global threat, driven by climate change and rising international travel.
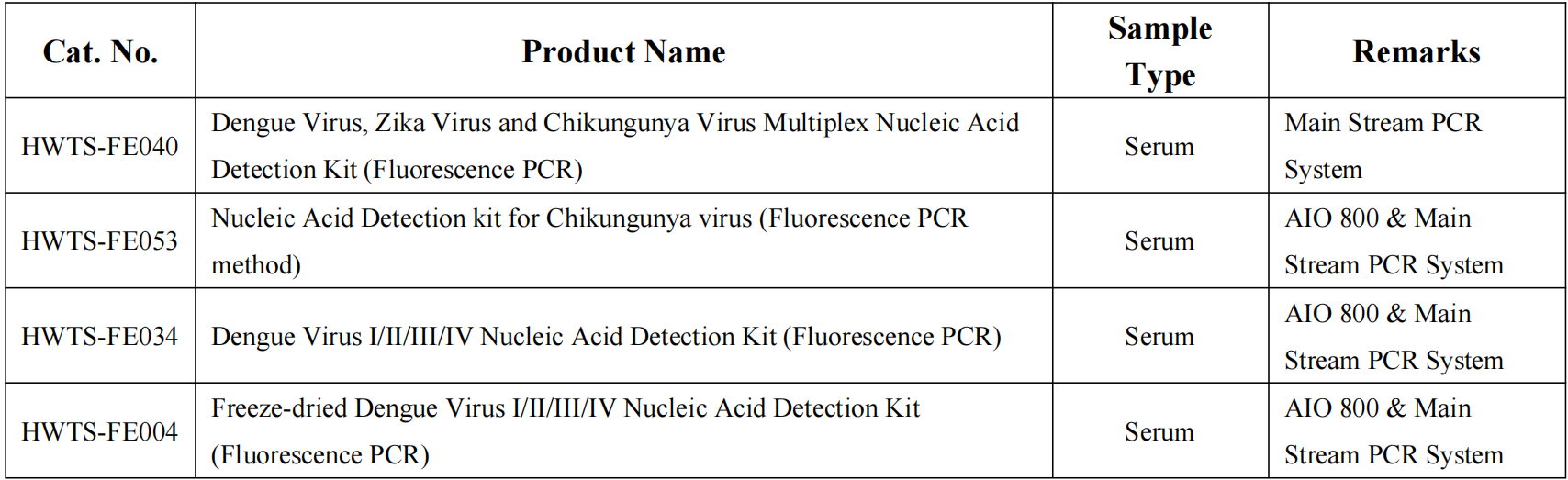
Recent data show both viruses intensifying across multiple regions. In the Western Pacific, Cambodia reported 4,338 dengue cases (as of March 10, 2026), Timor-Leste saw a tenfold surge in February 2026 compared to the previous year, and Sri Lanka recorded 5,724 cases in February alone — its highest monthly total. Singapore continues to see DENV-2 as the dominant serotype.
In the Americas, 18 countries have ongoing CHIKVD outbreaks. As of March 2026, the region recorded 32,529 cases and 9 deaths, with Brazil hardest hit. Local transmission has re-emerged in areas previously free of the virus for years, including Saint Lucia, which reported its first locally acquired case since 2021. Suriname is facing a rapidly escalating outbreak (1,357 confirmed cases, 1 death; true infections may exceed 4,000). In Africa, outbreaks continue in Kenya, Madagascar, Somalia, and Seychelles — prompting a CDC Level 2 travel alert for Seychelles. Europe has seen limited cases (14 in mainland France and Corsica), but climate change is expanding the habitat of the tiger mosquito northward.
These trends highlight an urgent priority: diagnostic capacity. Without accurate, timely testing, outbreaks go undetected until they escalate. Robust diagnostics - from rapid tests to molecular assays - are essential for case confirmation, patient management, serotype surveillance, and breaking transmission chains. In response, China reclassified both dengue and chikungunya as Category B notifiable diseases (effective April 1, 2026), mandating stricter laboratory reporting. Chinese authorities assess that in 2026, the risk of imported cases triggering local outbreaks is higher than in previous years. As temperatures rise and travel rebounds, investing in diagnostic readiness is the key to shifting from reactive outbreak control to early, effective prevention.
Macro & Micro-Test Integrated Solutions for Mosquito-Borne Viruses
To support the full spectrum of dengue and chikungunya control strategies, Macro & Micro-Test provides an integrated diagnostic portfolio covering rapid screening, molecular confirmation, and genomic surveillance.
These solutions enable healthcare institutions and laboratories to respond effectively across different stages of outbreak management, from frontline screening to advanced epidemiological research.
Scenario 1: Rapid Screening and Targeted Surveillance
Application: Fever clinics and primary healthcare facilities, Community screening in outbreak regions, Port health and border quarantine stations.
Dengue NS1 Antigen:Enables early detection within 1–3 days after symptom onset, with results available in 15 minutes.
Dengue Virus IgM/IgG Antibody: Supports determination of primary versus secondary infection.
Dengue NS1 Antigen + IgM/IgG Dual: Simultaneous antigen and antibody detection, covering both early and later stages of infection.
Chikungunya Virus IgM/IgG Antibody: Enables rapid assessment of recent infection or prior exposure.
Scenario 2: Precision Diagnosis and Emergency Response
Application : Hospital clinical laboratories, National and regional CDC laboratories, Mobile laboratories for outbreak response.
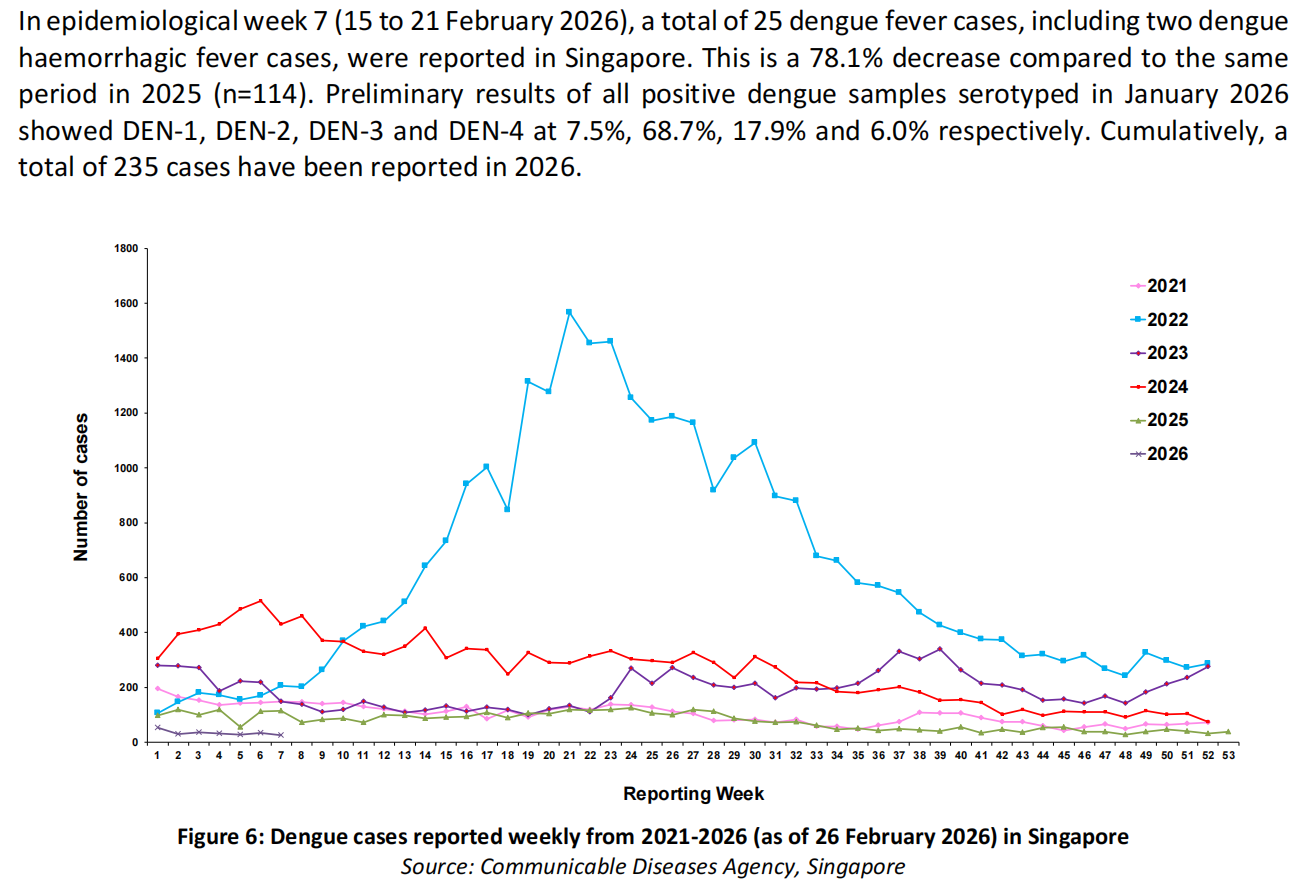
Dengue Virus I/II/III/IV Nucleic Acid: Detects and differentiates four dengue serotypes low to 500 copies/mL.
Lyophilized Dengue Virus PCR Kit: Enables room-temperature transportation and rapid deployment.
Dengue/Zika/Chikungunya Multiplex: Simultaneous detection of three major arboviruses,enablingefficient differential diagnosis during complex outbreaks.
Chikungunya Virus PCR Detection Kit: Suitable for early infection and low viral load cases with LoD of 200 copies/mL.
Compatible with the AIO 800 Fully Automated Sample-to-Answer System, minimizing manual operation and contamination risk
Scenario 3: Genomic Surveillance and Viral Lineage Analysis
Application: National reference laboratories, Public health research institutions, Academic and epidemiological research centers
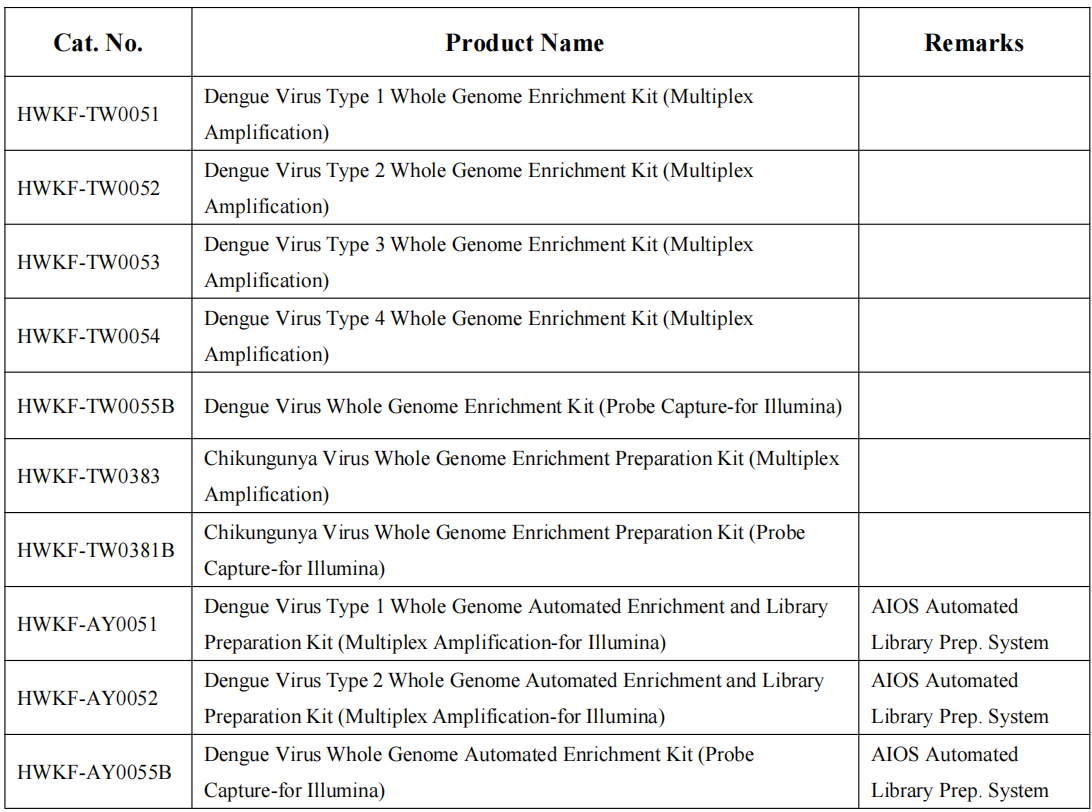
-Supports virus origin tracing and outbreak investigation
-Enables mutation monitoring and evolutionary analysis
-Flexible workflows for both manual and automated sequencing pipelines, enabling increased throughput and reproducibility.
Macro & Micro-Test Mosborne Disease Solutions: Accurate, Reliable, Comprehensive
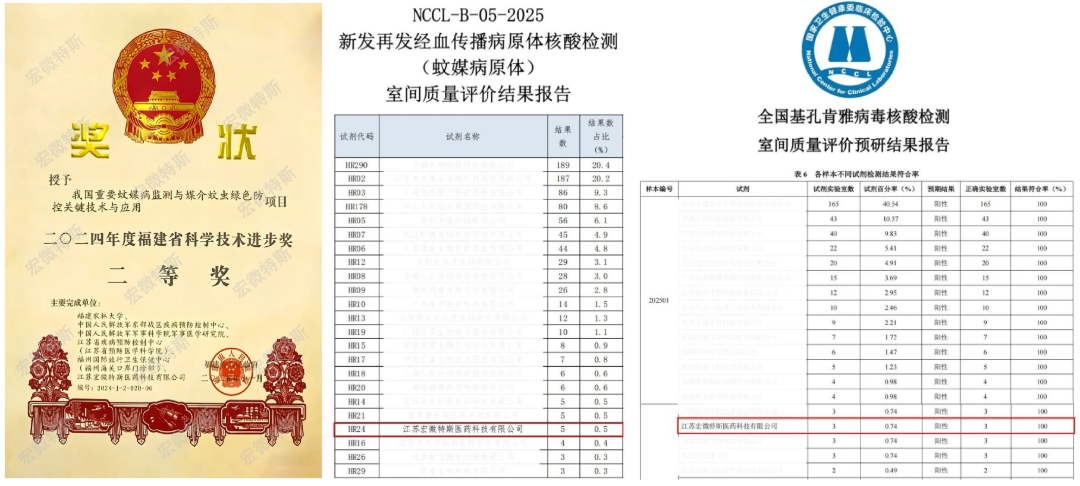
In a national external quality assessment (EQA) for chikungunya virus involving core laboratories across China, Macro & Micro-Test achieved 100% pass rate in both sensitivity and specificity — all samples were correctly identified with no false positives or false negatives, fully validating the robustness and reliability of the solution in real-world clinical and surveillance settings.
Macro & Micro-Test provides a complete diagnostic workflow for mosquito-borne virus detection, supporting every critical stage of outbreak management:
Rapid screening for frontline healthcare settings
Molecular confirmation for accurate diagnosis
Whole-genome analysis for epidemiological surveillance
With high-performance assays, flexible workflows, and automation ready platforms, the solution empowers laboratories and public health systems to strengthen preparedness and response against emerging arboviral threats worldwide.
�� Contact us for product details: marketing@mmtest.com
Post time: Apr-09-2026